 |
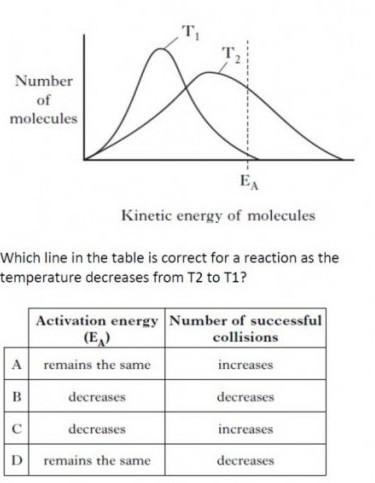
You should be familiar with this type of diagram. The particles (atoms or molecules) of solids liquids and gases are constantly in motion. The velocity with which a particle moves is a measure of its kinetic energy. The faster the speed, the greater the kinetic energy. For any substance, the particles making up the substance will be have a range of kinetic energy which can be represented by the type of curve in the question (Maxwell-Boltzmann distribution).As the temperature is increased, the kinetic energy of the particles increases (on average). Those particles with kinetic energy greater than the activation energy have the potential to react if a collision occurs. (Not all collisions result in a reaction as the colliding particles must also collide with favourable collision geometry). Consequently, if a greater proportion of particles have sufficient energy, when they collide more of the collisions will be successful. Activation energy is a characteristic of the reaction itself. It remains the same regardless of the temperature. Only a catalyst can alter (reduce) the activation energy.
|


