Click the QR code for MC Questions. Written questions, use your jotter, date & heading: Ex 1_3B Periodicity. |
 |
 |
| 1. | |
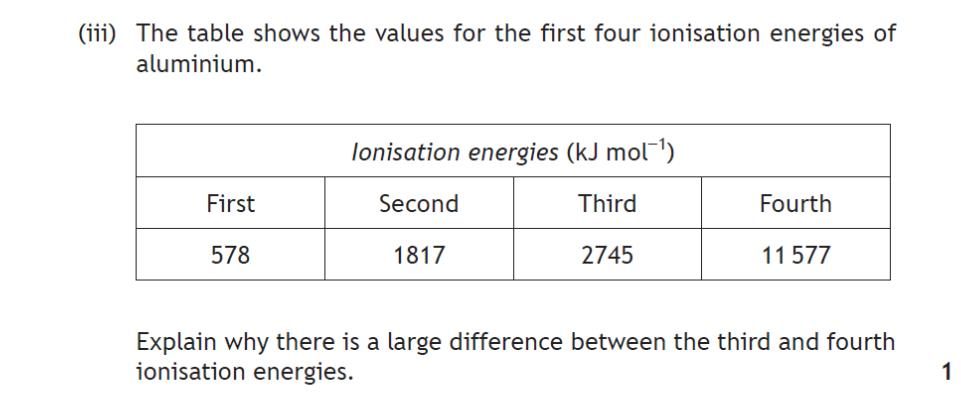
 1 mark for this question. |
|
| 2. | |
 2 marks for this question. |
|
 1 mark for this question. |
|
| 3. | |
 |
|
 |
|
 |
|
 |
|
 |
|
| 4. | |
 |
|
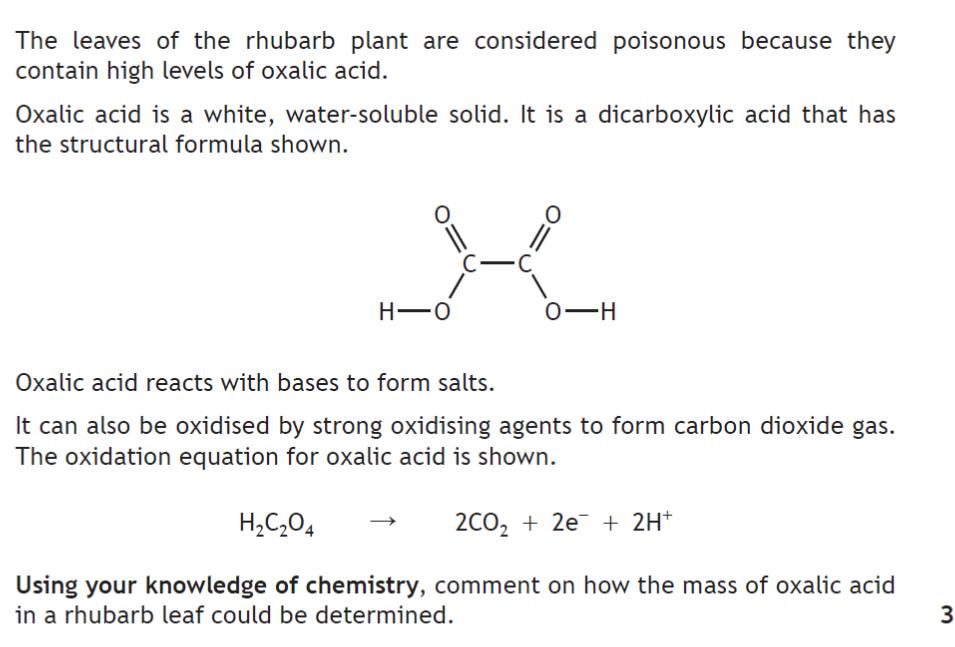
| 5. | |
 |
Higher Chemistry Unit 1 Consolidation Exercises
Hyndland Chemistry Department