 |
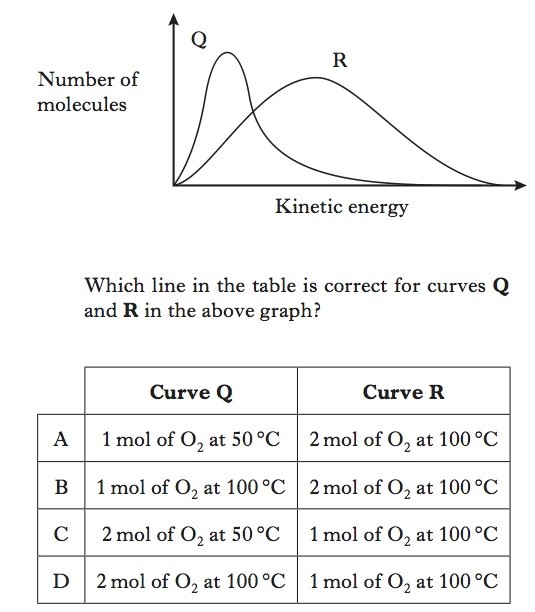
You should be familiar with this type of diagram.
The particles (atoms or molecules) of solids liquids and gases are constantly in motion. The velocity with which a particle moves is a measure of its kinetic energy. The faster the speed, the greater the kinetic energy. For any substance, the particles making up the substance will be have a range of kinetic energy which can be represented by the type of curve in the question (Maxwell-Boltzmann distribution).This question is comparing Q and R. Two features are to be compared, the energy and the quantity of particles. The energy is easiest: it is clear that in scenario R, more particles have more energy. This should allow you to reduce your choices to two options.As regards the quantity, the area under the curve indicates the quantity of particles. This is a little more difficult to tell from the graph, but nevertheless, R would seem to have a greater quantity of particles. |