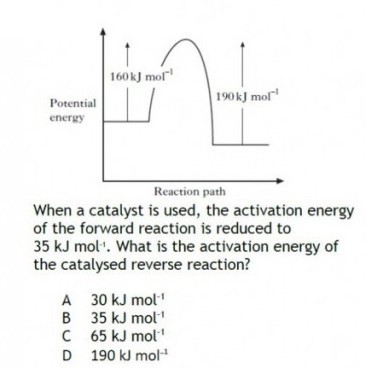
 |
This question can be a little tricky.
A number of approaches are possible. Probably it is easiest to recognise that the activation energy of the reverse reaction is 30 kj mol-1 greater than the forward reaction – this is because the enthalpy of the product (for the forward reaction) is 30 kj mol-1 lower than that of the reactant. That doesn’t change with a catalyst; a catalyst will reduce the Activation Energy (EA), but will not change the enthalpy change (or enthalpy of the chemicals involved). |

